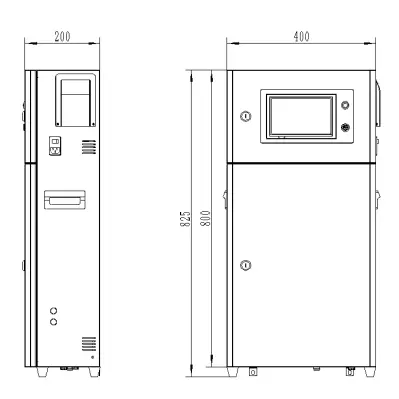
It seems we can’t find what you’re looking for. Perhaps searching can help.
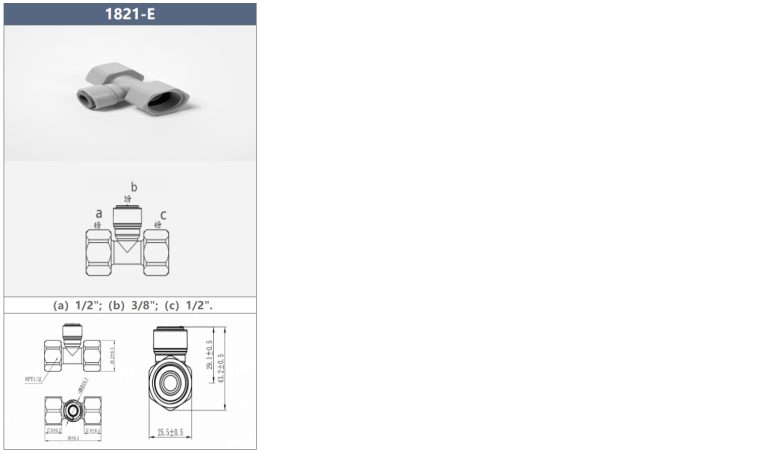
apakah perlengkapan pvc cocok dengan pipa abs
“Fitting PVC serbaguna untuk kompatibilitas sempurna dengan pipa ABS.” Potensi Masalah Kompatibilitas Antara Perlengkapan PVC dan Pipa ABS Dalam proyek perpipaan, penting untuk memastikan bahwa semua komponen kompatibel untuk menghindari potensi masalah di kemudian hari. Salah satu pertanyaan umum yang muncul adalah apakah fitting PVC cocok dengan pipa ABS. Meskipun PVC (polivinil klorida) dan ABS…